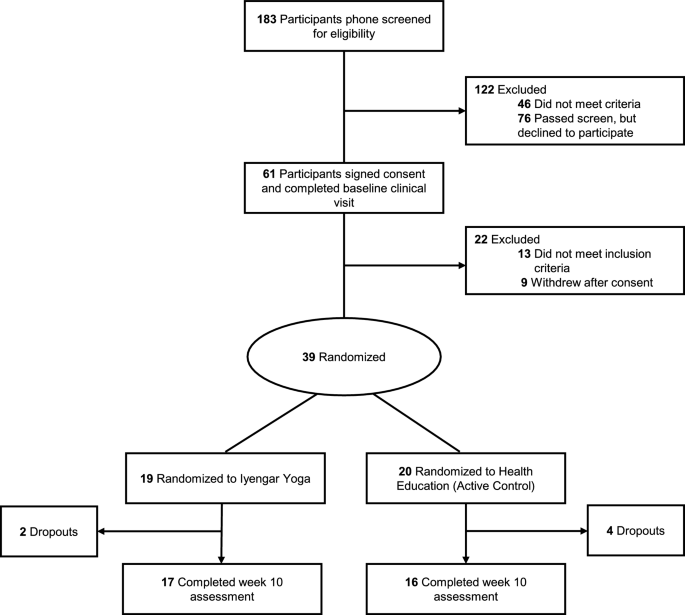
Study participants
This single-site RCT recruited 39 community-dwelling participants at the Medical College of Wisconsin (Milwaukee, Wis.) between October 2021 and December 2023, as part of a mechanistic feasibility trial titled “Iyengar Yoga to probe PGD neurobiology” (ClinicalTrials.gov #NCT05026827).
Inclusion criteria included individuals aged ≥ 40 years who had experienced the death of a loved one at least 6 months prior to enrollment and scored 25 or higher on the Inventory of Complicated Grief (ICG) scale29.
Exclusion criteria included a lifetime history of bipolar, psychotic, or neurological disorders (e.g., stroke, dementia, severe head injury), acute suicidality (assessed using Columbia Suicide Severity Rating Scale or clinician judgement), substance use disorders in the past three years, MRI contraindications, delirium/unstable medical conditions (i.e., Cumulative Illness Rating Scale – Geriatrics version (CIRS-G) score of 4 in any organ category), serious back, joint, or neck injuries within the past 3 months, or current yoga practice. All participants had intact basic and instrumental activities of daily living and at least ten years of formal education.
Comorbid depression, anxiety, and trauma- and stressor-related disorders were not exclusions. Antidepressants, low-dose benzodiazepines, and psychotherapy were allowed, provided they were stable for at least 4 weeks prior to enrollment and during the study.
All participants provided written informed consent. The study was approved by the MCW Institutional Review Board (IRB) and registered with ClinicalTrials.gov (Identifier: NCT05026827) before recruitment began. All methods were performed in accordance with the MCW IRB guidelines and regulations.
Study procedures
Phone screen
Potential bereaved participants were initially phone screened for general inclusion/exclusion criteria. Those who scored 3 or more on the Brief Grief Questionnaire (BGQ)30 were scheduled for the study baseline visit.
Study baseline visit
All eligible participants were assessed for their capacity to consent using the UCSD Brief Assessment of Capacity to Consent31 adapted for this study.
After providing written informed consent, participants completed a comprehensive clinical assessment, including sociodemographic, medical, psychiatric, and medication histories, a neurological examination, a thorough assessment of inclusion/exclusion criteria, and the Structured Clinical Interview for DSM-5 Research Version32. According to DSM-5-TR, a diagnosis of PGD requires that the bereavement occurred at least 12 months prior to assessment. Thirty-six of the 39 participants met this duration criterion and were administered the Structured Clinical Interview for Complicated Grief (SCI-CG) to assess PGD diagnosis33.
Participants completed self-report clinical scales, including the 19-item Inventory of Complicated Grief (ICG) scale29 for overall grief, the Patient Health Questionnaire-9 (PHQ-9)34 for depressive symptoms, and the 21-item Yearning in Situations of Loss (YSL)-Bereaved35, the Impact of Event Scale-Revised (IES-R)36, Grief-Related Avoidance Questionnaire (GRAQ)37 and 20-item UCLA loneliness scale-version 3 (UCLA-3)38 for grief-specific symptoms.
Additionally, study personnel administered the Montreal Cognitive Assessment (MoCA)39, and CIRS-G40. The relationship of the deceased and time since the loss (TSL) were collected, and psychotropic medication use and psychotherapy attendance were documented using the Antidepressant Treatment History Form (ATHF)41. Participants also completed the Duke Social Support Inventory42, Lubben Social Network43, and Community Health Activities Model Program for Seniors (CHAMPS)44 physical activity questionnaires.
Randomization
After baseline assessments, eligible participants were randomly assigned to either IY or HE group interventions using a computer-generated randomization scheme with block randomization to assign participants to a group (i.e., 2:2 or 3:3 ratio to a group) ensuring an equal number of participants in each group. See supplement for details.
IY intervention
Participants attended 60-min classes held once weekly for 10 weeks. Classes were taught by expert IY instructors (LM, KC) with certification by the IY National Association of the United States and > 10 years of experience teaching IY, including to older adults. Each wave comprised of 10 classes; each 60-min class included (1) breathing practices (pranayama) (~ 5-min), (2) gentle postures (asana) (~ 40-min), (3) mindfulness meditation (~ 10-min), concluding with (4) seated centering (5-min). After each class, participants were given home poses to practice over the following week. The participants were asked to practice breathing exercises and poses for at least 15 min, five days a week, and asked to record their home practice duration. IY instructors also completed weekly attendance and fidelity logs.
Health education (socialization active control) group
HE sessions were held weekly for 10 weeks. Each session consisted of a 50–55-min video viewing (two ~ 25–30-min videos from the Great Courses lectures, used with permission under a group viewing site license for academic purposes; see supplementary methods S2 and Table S1), followed by ~ 5–10 min discussion facilitated by a study team member (KRH or AW). All waves viewed the same 20 videos of lectures in the same sequence. All videos were thoroughly vetted by the study team prior to use, to ensure that the lectures did not include information relevant to the active intervention arm (IY components) or information regarding grief, exercise, or depression. The individual lectures included were from the following courses: (1) The Aging Brain, (2) Lifelong Health: Achieving Optimum Health at Any Age, (3) Your best brain, and (4) Sensation, perception, and the aging process45,46,47,48.
Longitudinal assessments
After randomization, participants completed the ICG, PHQ-9, and YSL once every 2 weeks for 10 weeks. Self-report (IES-R, GRAQ, UCLA-3) and clinician-administered (CIRS-G, ATHF) scales were completed at weeks 4 and 10.
Outcomes
See supplement (Methods S3) for details on feasibility and yoga fidelity assessments.
IY instructor fidelity
Two certified IY instructors (LM and KC) taught all classes. The PI (JSG) and co-investigator (KRH) briefed the instructors regarding PGD, the study design, and the IY intervention before the first wave. IY classes were videotaped, and 10% of the recordings were randomly selected and evaluated by an outside expert using a fidelity checklist. The IY instructors also met with a study investigator (JSG or KRH) at least once a month to ensure consistency across classes.
Other feasibility measures
Retention and attendance
Session participation in both groups was documented. Weekly attendance logs (for both IY and HE) and IY home practice logs were collected after each class.
Satisfaction
Participant satisfaction (enjoyment, perceived benefit, instructor quality, getting to know peers, common bond with peers) was assessed at week 10 using a Likert scale from 0 (not satisfied) to 10 (most satisfied)16.
Expectancy
The Opinions About Treatment (OAT) rating scale15,49 was adapted to gauge expectancy for the two interventions and completed at baseline. Participants self-rated four items on a 7-point scale. Summing the ratings on IY and on HE produced two scores (ranging from 4 to 28) per participant, corresponding to IY and HE intervention expectancy, respectively. Higher scores indicated greater expectations of treatment benefit (see supplementary material for details).
Safety monitoring
Adverse events (AEs) were documented weekly using an AE form, with each event evaluated by the PI. If a participant endorsed a possible serious adverse event (SAE) or needed immediate intervention (e.g., suicidal ideation), staff immediately contacted the PI.
Clinical
The primary outcomes were changes in ICG and PHQ-9 scores over 10 weeks. Secondary outcomes included changes in YSL, IES-R intrusion subscale, GRAQ, and UCLA-3 scores. Additionally, response (defined as ≥ 30% reduction in ICG score) and remission (ICG score < 20 at week 10) were documented.
Statistical analysis
Descriptive statistics, means and standard deviations for quantitative measures or frequencies and percentages for each level of nominal characteristics, are calculated for each intervention group (IY, n = 19 and HE, n = 20) using the study baseline data. Two-sided t-tests for quantitative data and Fisher’s exact tests for categorical or nominal data are calculated to determine whether the IY and HE groups were dissimilar at baseline. Participant satisfaction and expectancy rating scales were compared between groups (i.e., IY vs. HE) using the two-sample t-test, and behavior modification at week 10 with Fisher’s exact tests.
For clinical outcomes, mean changes (i.e., baseline—Week 10) and 95% confidence intervals are calculated separately for each intervention group. The difference in mean changes between the two groups (IY vs. HE) are also calculated, along with its 95% confidence interval. Hedges’ g effect size is calculated to quantify the magnitude of change within each intervention group and between groups.
link